Glioma is a highly heterogeneous, aggressive primary brain tumor. The current standard of care for patients diagnosed with glioma includes performing a maximum safe extent resection of the tumor, undergoing radiotherapy over a 6-week period, and receiving concomitant systemic therapy with the alkylating agent TMZ (1). In addition to this, treatments such as immunotherapy, targeted precision therapy, electric field therapy, and supportive care have resulted in some improvement in the survival duration of glioma patients. However, these patients typically experience a poor prognosis and low quality of life, with only a 4-5% 5-year survival rate (2). Furthermore, cognitive and focal neurological deficits can significantly impact long-term survivors of brain tumors, regardless of tumor histology and grading (3).
The genesis of the scientific inquiry into RCD is often attributed to Karl Vogt’s discernment of cellular demise in toads in 1842. A significant leap in the conceptual framework occurred in 1972 when Kerr, Wyllie, and Currie introduced the term “apoptosis” to the research lexicon, propelling the study of RCD to the scientific forefront (4). The discovery by Sulston and Horvitz in 1976, utilizing the model organism C. elegans, was instrumental in unearthing the genetic underpinnings of apoptosis and revealing a predetermined fate for approximately 13% of somatic cells during embryogenesis (5). Over the past decade, NCCD has meticulously curated a taxonomy of cell death modalities, based on distinguishing cellular morphology, biochemistry, and functional outcomes. The dichotomy between cell death pathways lies within two realms: accidental pathways triggered by external factors such as mechanical, chemical, and physical stressors that lead to cellular demise when they surpass the cell’s compensatory capacities; and RCD, which represents an inherent and orchestrated cessation of cellular function mediated precisely by genetic regulation to maintain internal homeostasis. This gene-directed cessation in a physiological context is also known as programmed cell death (PCD) (6). Diverging from the unregulated nature of accidental cell death (ACD), RCD involves a meticulous network of signaling cascades and molecular machinery. The exploration of RCD has revealed numerous novel mechanisms with profound connections to human pathologies, expanding beyond apoptosis to encompass diverse processes such as autophagy-dependent cell death, ferroptosis, lysosome-dependent cell death, mitochondrial permeability transition (MPT)-driven necrosis, necroptosis, pyroptosis, anoikis, NETosis, among others (7). Each mechanism reflects a unique aspect of the cell’s ability to maintain physiological integrity and their ongoing elucidation holds significant promise in enlightening the pathogenesis of various diseases and refining therapeutic strategies.
RCD, a significant pathway in the cell death process, exhibits dual effects in central nervous system tumors, particularly glioma. On one hand, dysregulated apoptotic pathways in gliomas can contribute to the anti-apoptotic properties of tumor cells, thereby facilitating tumor growth and resistance to treatment (8, 9). Over-expression of anti-apoptotic proteins, such as members of the Bcl-2 family, by glioma cells potentially inhibits the activation of RCD (10, 11). Moreover, the abnormal regulation of RCD pathways can make glioma cells resistant to conventional therapies such as RT and chemotherapy (12). However, in certain scenarios, RCD can have an inhibitory impact on glioma. Studies have indicated that by enhancing RCD in glioma cells, tumor volume can be reduced and treatment efficacy can be improved (13, 14).
Despite the progress made in the realm of RCD, the journey toward its clinical application remains challenging. To date, therapeutic strategies harnessing apoptosis have only yielded a limited number of approved pharmacological agents for human disease management. No agents that intentionally inhibit apoptosis have been established for clinical use. An example of an approved therapy is Venetoclax, a BCL2 inhibitor which has demonstrated efficacy as a monotherapy or in combination regimens for treating chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), and acute myeloid leukemia (AML) (15–17). Furthermore, the caspase inhibitor Emricasan garnered accelerated attention from the U.S. FDA in 2016 for its focus on non-alcoholic steatohepatitis (NASH); nevertheless, the clinical outcomes have exhibited variability, underscoring the unpredictable nature of current interventions (18, 19). Looking ahead, a comprehensive investigation into the molecular intricacies of cell death, particularly within the context of the interconnected networks among various RCD pathways, is expected to stimulate innovative breakthroughs. It is through such a thorough understanding of the interplay between these pathways that novel therapeutic strategies can be developed, paving the way for their translation from laboratory research to practical application in medical treatment. The emphasis on these mechanistic insights holds great promise for unveiling new opportunities for clinical intervention and represents a significant advancement in harnessing RCD for disease management.
Overall, the regulation of RCD in glioma involves intricate signaling pathways and regulatory factors. Its influence on glioma depends on the complex interplay of multiple factors, including tumor cell characteristics, the tumor microenvironment, and treatment modalities (20, 21). Thus, this review provides a concise overview of the role of RCD in the onset, treatment, and prognosis of glioma. It focuses on several significant apoptotic pathways including autophagy-dependent cell death (22, 23), anoikis (24), ferroptosis (25, 26), cuproptosis (27), pyroptosis and immunogenic cell death (Figures 1, 2; Table 1). We propose strategies to optimize and coordinate traditional or novel approaches for glioma treatment based on these pathways. This comprehensive understanding of RCD in glioma will not only enhance readers’ comprehension of its underlying mechanisms but also serve as a theoretical foundation for developing more effective treatment strategies.
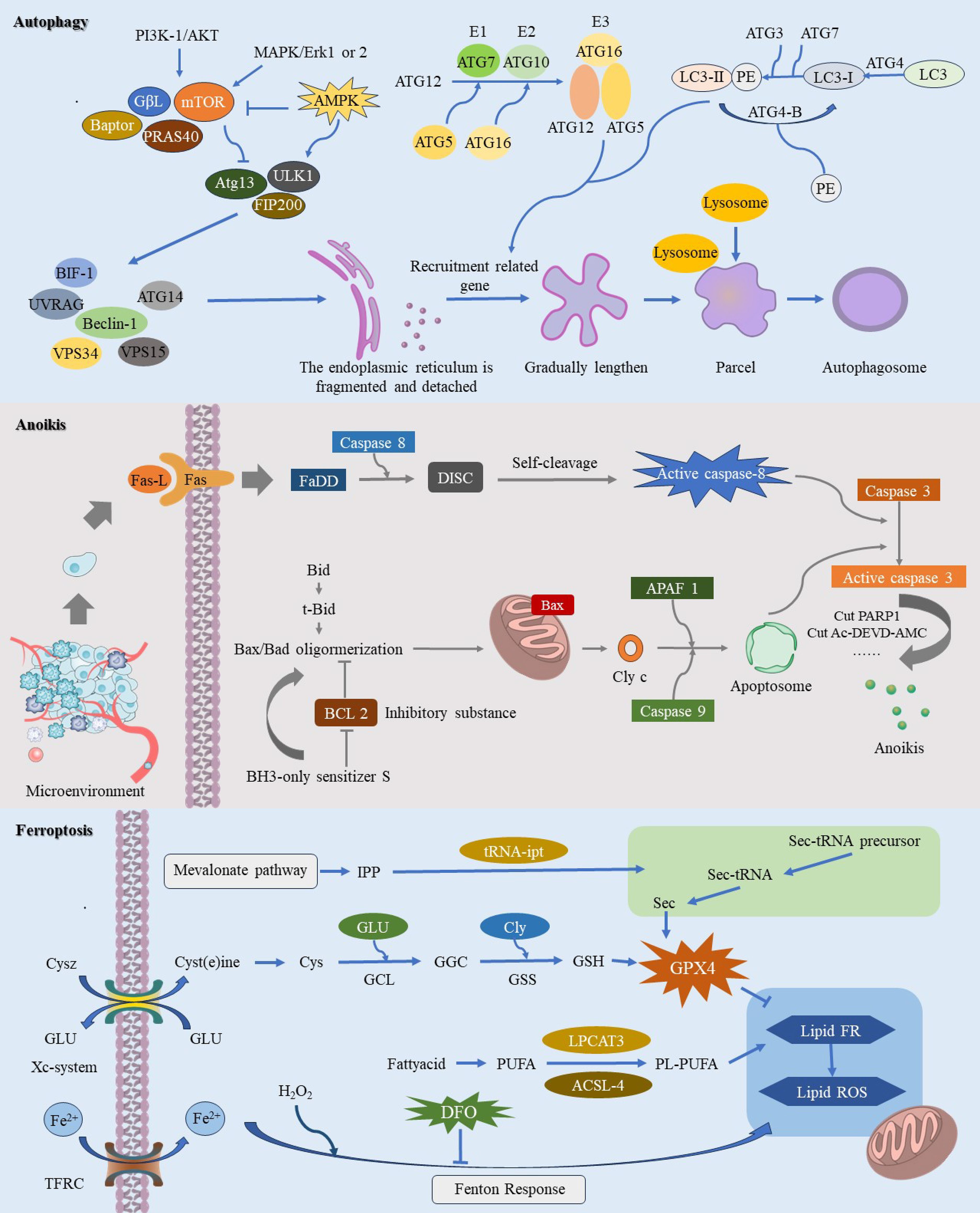
Figure 1. This image provides a comprehensive illustration of the molecular mechanisms underlying three pathways of regulated cell death (RCD): autophagy-dependent cell death, anoikis, and ferroptosis. Autophagy-dependent cell death is characterized by the crucial role of autophagy in degrading essential cellular components, ultimately leading to cell demise. During autophagy-dependent cell death, continuous autophagic activity undermines cellular structure and function, resulting in eventual cell death. Anoikis is primarily initiated when cells dissociate from the extracellular matrix, causing them to lose vital survival signals. This detachment disrupts integrin and other adhesion molecule signaling pathways, leading to the downregulation of survival pathways such as PI3K/Akt and Ras/MAPK, while upregulating pro-apoptotic factors like Bim and Bid. The resulting alterations in mitochondrial membrane potential facilitate the insertion of pro-apoptotic proteins Bax and Bak into the mitochondrial membrane, which then releases cytochrome c and activates downstream apoptotic caspases, ultimately driving cell apoptosis. The process of ferroptosis is initiated by elevated intracellular iron levels, which amplify the Fenton reaction and enhance ROS production. This process triggers lipid peroxidation of polyunsaturated fatty acids in the cell membrane, leading to oxidative stress and compromising membrane integrity, ultimately resulting in cell death.
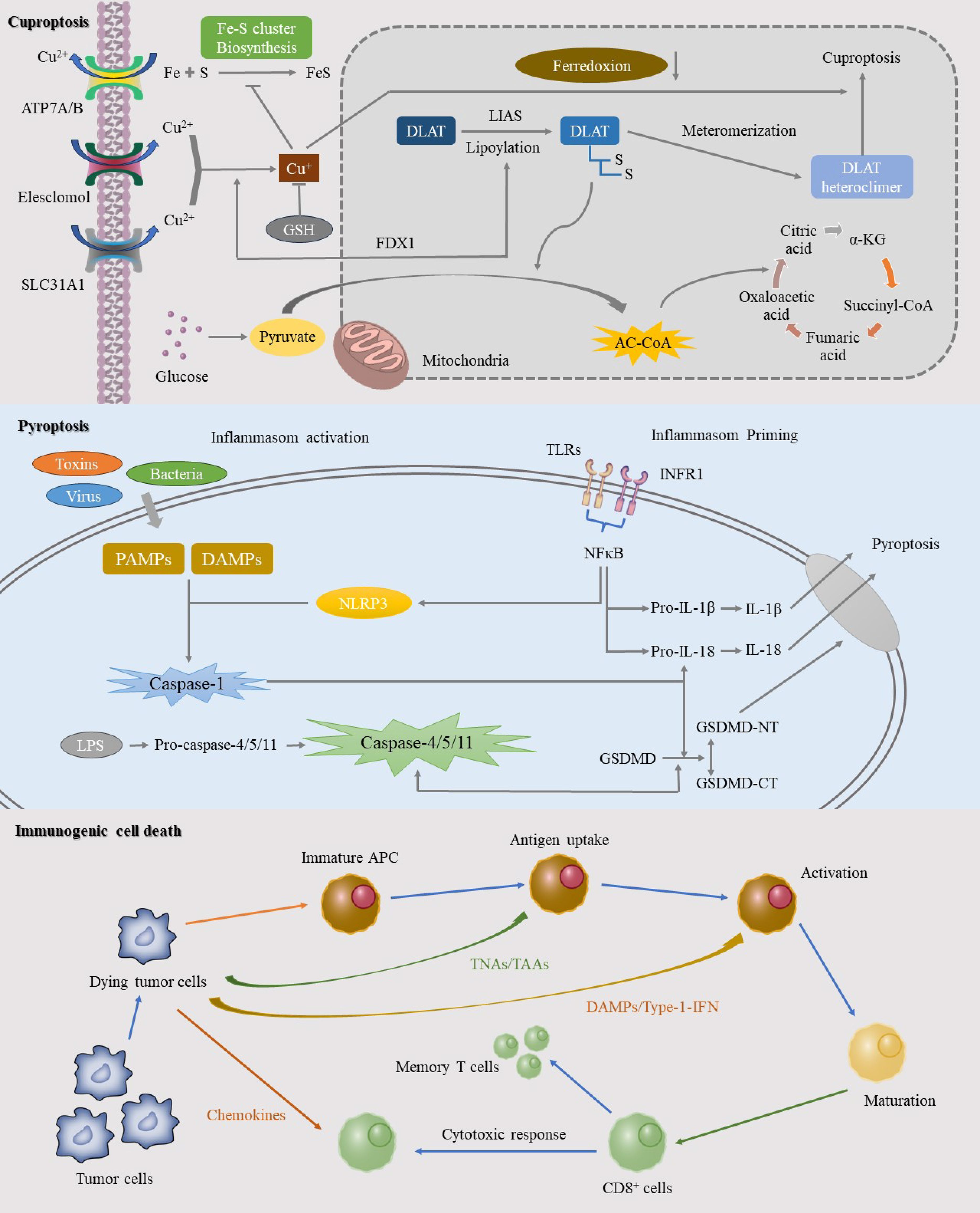
Figure 2. This image elucidates the specific mechanisms of three significant RCD pathways: cuproptosis, pyroptosis, and ICD. Cuproptosis, a copper-dependent form of cell death, is initiated by the intracellular accumulation of copper ions. The mechanism involves the binding of copper ions to thioredoxin cluster proteins, leading to protein aggregation and mitochondrial dysfunction. Pyroptosis, another RCD process, is mediated by inflammasome activation in response to pathogens or intracellular danger signals, resulting in a robust inflammatory response. ICD represents an RCD pathway that triggers a potent anti-tumor immune response. This process involves the release of specific death signals and DAMPs that activate the immune system.
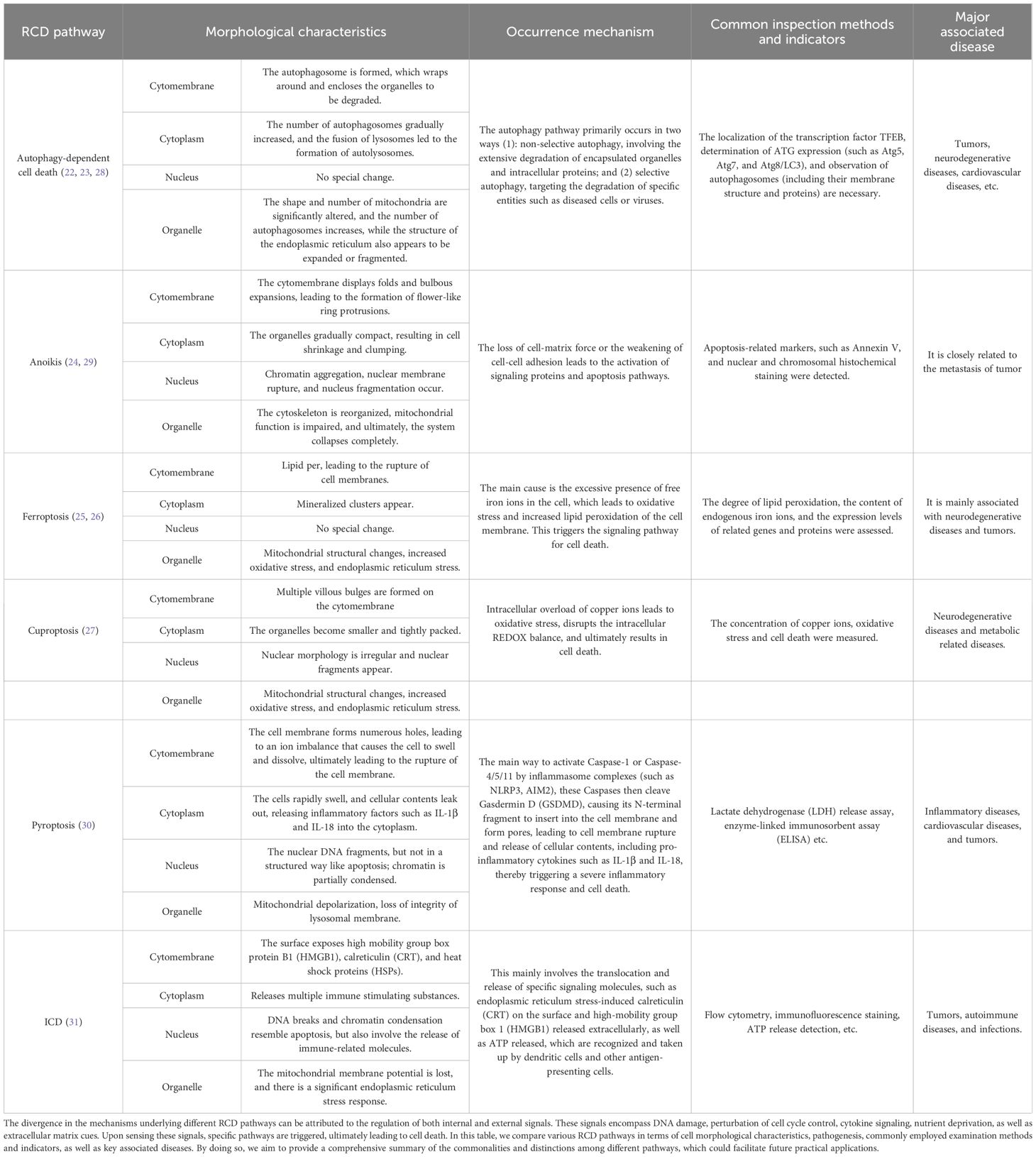
Table 1. Comparison of distinct aspects of RCD pathways.
2 Glioma and glioma immunotherapyGliomas represent 40-60% of primary central nervous system tumors in adults and are the most prevalent primary intracranial tumors in this population. According to the World Health Organization (WHO), gliomas are classified into four grades: grades 1 and 2 are low-grade, whereas grades 3 and 4 are high-grade (32). Despite advancements in glioma treatment over the past decades, challenges persist due to their unique location and the characteristics of “cold” tumors, which contribute to high recurrence rates, poor prognoses, and short median survival times (33).
The immune system fulfills three critical roles: immune surveillance, immune defense, and immune homeostasis, all of which are integral to tumor initiation, progression, metastasis, and response to treatment. Immune checkpoint inhibitors (ICIs) have revolutionized cancer therapy in recent years. However, gliomas, characterized as “cold” tumors, often exhibit poor or non-responsive behavior to ICIs, limiting the success of immunotherapy. Enhancing the immune microenvironment to convert “cold” tumors into “hot” ones may provide a new avenue for improving treatment responses (34, 35).
Bevacizumab, an FDA-approved monoclonal antibody targeting vascular endothelial growth factor (VEGF), inhibits tumor vascularization rather than directly targeting T cells (36). PD-1 inhibitors such as Pembrolizumab and Nivolumab have shown promise in clinical trials. PD-1, a receptor on T cells, interacts with PD-L1 to suppress T cell activity, aiding tumor immune evasion. Pembrolizumab disrupts this interaction, reactivating T cells to recognize and attack tumor cells (37, 38). Similarly, Nivolumab restores T cell functions by blocking the PD-1 receptor. Although a phase III clinical trial comparing Nivolumab plus RT to TMZ plus RT in newly diagnosed patients with non-methylated MGMT promoter gliomas did not achieve the primary endpoint of extending overall survival, it marked a significant step towards using Nivolumab with RT in glioma treatment, without identifying major safety concerns (39). Other immunotherapy strategies are also being explored. Oncolytic viruses (OVs) selectively replicate within and induce apoptosis in cancer cells while sparing normal tissues. A multicenter study using the oncolytic virus DNX-2401 in combination with an anti-PD-1 monoclonal antibody demonstrated safety and survival benefits (40). Dendritic cell (DC) vaccines like DCVax-L hold promise in clinical trials; these vaccines involve extracting and loading dendritic cells with tumor antigens in vitro before re-injecting them into the patient to stimulate an immune response (41).
The Nobel Prize in Medicine was awarded to James Allison and Tasuku Honjo six years ago for their groundbreaking work in immunotherapy (42). Current standard treatment for glioblastoma includes surgical resection combined with TMZ chemotherapy but remains insufficient. Recent studies indicate that RCD can play a synergistic role in anti-tumor immunotherapy, including for ICI-resistant tumors. Existing research highlights significant distinctions in RCD processes between low-grade and high-grade gliomas. In low-grade gliomas, apoptosis levels are relatively low, and the role of autophagy remains controversial, though these tumors generally exhibit better cellular survival activity. By contrast, high-grade gliomas demonstrate a higher resistance to apoptosis and possess a more intricate autophagy function, often accompanied by more extensive necrotic areas. These malignant tumors feature markedly different gene expression profiles, which in turn lead to varied treatment responses. Low-grade gliomas tend to be more responsive to radiation and chemotherapy, while high-grade gliomas exhibit a stronger resistance to these treatments (43). As immunotherapy advances, it becomes crucial to investigate the different pathways that either inhibit or activate various RCD processes, as well as their synergistic effects. Such exploration could pave the way for more effective targeted treatments for gliomas.
2 Regulated cell death and glioma2.1 Autophagy-dependent cell deathCoined by Christian de Duve and colleagues in 1963, the term “autophagy” describes the process by which cellular components, whether originating from within or outside the cell, are transported to lysosomes for degradation (44). Since the 1990s, research on autophagy has been greatly expanded due to its use as a model organism in yeast studies. Initial findings showed an accumulation of autophagosomes and lysosomes in dying cells, leading to the naming of “autophagic cell death” (45, 46). Autophagy plays multifaceted roles throughout disease progression, particularly in tumor. In the early stages of oncogenesis, autophagy acts as a guardian of genomic integrity and internal stability by regulating quality control and responding to oxidative stress. This helps prevent tissue damage, inflammation, and ultimately inhibits tumorigenesis and metastasis. However, during advanced stages of tumor, autophagy can serve as a source of nutrients that facilitate neoplastic growth, enhance immune evasion, and promote tumor advancement (47). Historically, reliance on morphological delineation has been the cornerstone of autophagy research, resulting in a gap in establishing causal relationships between autophagy and cell death. The present consensus among scientists categorizes autophagy’s involvement in cell death into three primary types: autophagy-related, autophagy-dependent, and autophagy-mediated cell death (48). Autophagy-dependent cell death is considered as a regulated form of cell death that relies on the autophagic apparatus and generally adheres to the following criteria (1): elevated autophagy flux occurring simultaneously with cell death (2), reversibility of cell death when autophagy is inhibited through pharmacological or genetic means (3), dependence on at least two components of the autophagic process, and (4) absence of concurrent alternative forms of cell death (6).
The notion of autophagy-dependent ferroptosis emerged amidst deepening investigations into ferroptosis, a form of cell death described in 2012 as iron-dependent and distinct from apoptosis, autophagy, and necrosis (49). However, the specificity of this classification has come under scrutiny. Because Minghui Gao et al. found that under the action of ferroptosis activator, autophagosomes gradually accumulated, and cells died because of some components of the autophagy mechanism, they named this death mode as autophagy dependent ferroptosis (50–52). This reevaluation highlights the dynamic and interconnected nature of autophagic processes and cell fate, keeping autophagy at the forefront of contemporary biomedical research.
As far as the current research on glioma is concerned, most of the research still stays on the acceleration or delay of the autophagy process. Celastrol, a triterpenoid compound derived from Tripterygium wilfordii, a traditional Chinese medicine, demonstrates potential anti-glioma effects. It activates the ROS/JNK signaling pathway while inhibiting the Akt/mTOR pathway, leading to G2/M phase arrest and triggering autophagy (53). Thiolidazine enhances P62-mediated autophagy by upregulating AMPK activity and the Wnt/β-catenin signaling pathway, thereby inhibiting glioma cell proliferation and migration (54).
In this case, there are still a few studies to explore the relationship between autophagy dependent cell death and the occurrence and development of glioma. Investigator Svenja Zielke established a rigorous criterion for autophagy-dependent cell death and carefully identified three effective agents - loperamide, pimozide, and STF-62247 - from a wide range of over 70 compounds that induce autophagy. These agents were observed to promote LC3B lipidation and puncta formation, which are hallmarks of autophagic activity. This promotes autophagic flux through interactions with essential autophagy proteins ATG5 and ATG3. Ultrastructural analysis revealed an abundance of autophagosomes and autolysosomes in cells treated with loperamide and pimozide. Additionally, these compounds induced a noticeable dephosphorylation trend in complex 1 of the mammalian targets of rapamycin (mTOR), indicating activation of autophagy. Consequently, these findings suggest that loperamide, pimozide, and STF-62247 could potentially trigger autophagy-dependent cell death in glioma cells, providing a novel paradigm for therapeutic intervention in glioblastomas (55). Separately, amentoflavone (AF), a polyphenolic compound endemic to the Selaginella species, exhibits a broad spectrum of biological activities including anti-inflammatory, anti-neoplastic, radioprotective, antioxidative, and neuroprotective properties. Experimental observations have documented conspicuous depletion in intracellular glutathione levels and mitochondrial membrane potential following AF exposure, concomitant with a surge in cellular iron concentrations, malondialdehyde, and lipid peroxidation. Subsequent inquiry has substantiated the capability of AF to inhibit neoplastic proliferation by instigating autophagy-dependent ferroptosis in vivo potentially attributable to the modulation of the AMP-activated protein kinase (AMPK)/mTOR signaling cascade (56). These insights could delineate unrecognized molecular conduits for curtailing tumor growth and reinforce the therapeutic potential of autophagy-centric interventions in tumor management.
2.2 AnoikisWhen a cell loses its normal connection to the stroma, it senses signaling abnormalities that trigger anoikis, leading to disruption of intracellular signaling and cell death. Ultimately, this results in the effective removal of displaced cells (29). Because of this characteristic, this RCD pathway has been named amnesiotic apoptosis. NCCD defines lost-nest apoptosis as a specific variant of intrinsic apoptosis induced by integrin-dependent anchoring loss (6).
In the case of tumors, the absence of anoikis can promote tumor cell survival and proliferation, thereby facilitating tumor development and drug resistance (57–59).
With advancements in bioinformatics, several researchers have developed predictive models based on genes associated with anoikis. Zhongzheng Sun et al. utilized nine genes related to anoikis to predict the survival rate and prognosis of glioma patients. They constructed a risk score and employed this model to forecast patient outcomes. Based on these findings, the possibility of exploring immunotherapy for glioma using genes and mechanisms correlated with anoikis was investigated (60). Dongdong Zhang analyzed the expression and survival of genes related to five types of anoikis, including ETV4, HMOX1, MYC, NFE2L2, and UBE2C. Additionally, a clinical prediction model was constructed (61). This work laid the foundation for subsequent progress in basic experimental research. The lncRNA ANRIL exhibited a positive correlation with glioma grade in glioma tissues regarding the induction of anoikis, and it was found to indirectly induce anoikis by inhibiting the anti-apoptosis gene Bcl-2 (62). In terms of the tumor microenvironment, MNX1 was found to be ectopically expressed in glioma cells and associated with glioma grade. This substance was observed to contribute to cell adhesion and potentially enhance the ability of glioma cells to evade anoikis by regulating the expression of its downstream molecule TrkB (63).
2.3 FerroptosisFerroptosis is a recently discovered novel form of RCD that is dependent on reactive oxygen species (ROS). The process of cell death is typically accompanied by significant accumulation of iron and lipid peroxidation (64). With the deepening of the understanding of ferroptosis, many studies believe that ferroptosis should be attributed to autophagy dependent ferroptosis, but due to the uniqueness of its cause and process, it is still discussed as a separate concept here.
Ferroptosis is intricately linked to various diseases and pathological processes, encompassing neurological conditions (e.g., Parkinson’s disease, Alzheimer’s disease) (65, 66), cardiovascular diseases (67), and liver diseases (68). The orchestrated process of ferroptosis in gliomas is primarily regulated by four pathways, including metabolism, the GPX4 pathway, the FSP1 pathway, and lipid metabolism (69). In the process of iron metabolism, cells absorb ferric ions (Fe3+) via the transferrin receptor TfR1, and subsequently reduce them to ferrous ions (Fe2+). The GPX4 pathway represents the classical pathway of ferroptosis. At the heart of this pathway is the lipid repair enzyme glutathione peroxidase 4 (GPX4), which plays a crucial role in inhibiting intracellular lipid peroxidation by degrading small molecule peroxides and some lipid peroxides using GSH as a substrate (70).
Current studies have provided evidence that various factors and pathways can either induce or inhibit ferroptosis in glioma. Strychnine, a weakly basic indole alkaloid derived from strychnine seeds, has demonstrated potent antitumor activity against multiple tumor types, including glioma (71). Recent investigations have revealed its ability to induce ferroptosis in glioma cells (72). TRIM7, which utilizes a K4-linked chain, directly binds to and ubiquitinates nuclear receptor coactivator 4 (NCOA4), thereby reducing NCOA4-mediated ferritin phagocytosis and subsequent ferroptosis in human glioblastoma cells (73). Ferroptosis exhibits both beneficial and adverse effects in the development and progression of glioma. Some studies have suggested that increased ferroptosis may compromise the efficacy of PD-L1 immunotherapy, while inhibiting ferroptosis remodels the immunosuppressive microenvironment and enhances the effectiveness of immunotherapy (74). Conversely, plumbagin has exhibited tumor growth inhibition by targeting the NQO1/GPX4 pathway-mediated ferroptosis, suggesting therapeutic implications for inducing ferroptosis (75). In summary, ferroptosis exhibits dual effects on glioma, influencing the efficacy of immunotherapy and potential therapeutic strategies. However, a comprehensive understanding of ferroptosis in gliomas remains elusive, necessitating further research to identify key molecular targets and mechanisms.
2.4 CuproptosisSimilar to ferroptosis, cuproptosis is a copper-dependent and unique cell death (76). A recent study suggests that cuproptosis is an independent form of RCD and is highly associated with mitochondrial respiration and the lipoic acid (LA) pathway (77), and plays a key role in tumor cell proliferation, metastasis, and drug resistance.
Studies on cuproptosis in glioma have mainly relied on bioinformatics and gene sequencing techniques for which there are limited experimental validations available. Due to the restricted predictive value of individual biomarkers, researchers have incorporated multiple biomarkers into models to predict the behavior and progression of glioma accurately. For example, Bo Chen et al., identified a signaling pathway along with 13 ligand-receptor pairs such as ICAM8, ITGAX, ITGB1 ANXA2-FRR1 etc., constructing an activity score based on genes associated with cuproptosis while ensuring its stability through machine learning technology (78). Similarly, Lin Wang et al. identified 10 lncRNAs associated with cuproptosis that target 7 resistance genes (FDX1, LIAS, LIPT1, DLD, DLAT, PDHA1, and PDHB) and 3 sensitization genes (MTF1, GLS, and CDKN2A). They developed a prognostic risk model based on these cuproptosis-related lncRNAs which accurately predicts the tumor microenvironment status (79). Notably, FDX1 has been found to be highly expressed in gliomas and is primarily involved in lipid acylation of tumor proteins and cuproptosis, which significantly affects the prognosis of low-grade gliomas (80). Methylation of FDX1 is believed to promote malignant behavior in glioma. Functional experiments further revealed that the target gene C-MYC enhances the proliferation and invasion of glioma cells through YTHDF1 and FDX1 methylation, possibly due to aberrant copper ion behavior in mitochondria (81). Although existing studies are insufficient to establish the clinical value of cuproptosis-related mechanisms in glioma development and progression, multiple potential factors indicate that cuproptosis, like other RCD modalities, warrants further investigation and can profoundly impact glioma research.
2.5 PyroptosisPyroptosis, a newly identified form of RCD, plays a pivotal role in the body’s defense against pathogens. This mechanism is distinguished by specific morphological changes, including cell swelling, vesicle formation, membrane perforation, and eventual cell lysis. Unlike ferroptosis and autophagy, cell pyroptosis is mediated by proteins of the gasdermin (GSDM) family, which cause progressive cell swelling until membrane rupture. This process is tightly linked to inflammatory cascades and immune responses and can be categorized into classical and non-classical types based on the activation of caspase-1 (82).
To date, most investigations into pyroptosis in glioma have been conducted at the bioinformatics level, with limited experimental and clinical validation. Guilong Tanzhu and colleagues developed a prognostic model leveraging lncRNAs associated with pyroptosis to predict glioma patient outcomes. They further analyzed molecular alterations and immune infiltration across different risk groups, constructing a lncRNA-miRNA-mRNA regulatory network grounded in prognostic gene risk scores (83). Hanzhang Liu et al. identified nine key differential genes by examining 523 low-grade glioma (LGG) and 1,152 normal tissue samples. Their findings indicate that these genes are instrumental in LGG progression and tumor immunity, potentially benefiting prognosis and immunotherapy (84). Caspase-6 (CASP6), a critical protein in the pyroptosis pathway, presents fluctuating relevance in glioma prognosis. Kai Guo et al. conducted an extensive analysis of four bioinformatics data routes, extracting pyroptosis-related differentially expressed genes (PRDEGs) from 81 data sets within the GeneCards database. Their research revealed CASP6 overexpression in glioma, primarily implicating it in immune responses and antigen processing. CASP6 expression was inversely correlated with overall survival and disease progression in glioma patients (85).
In summary, bioinformatics analyses suggest a significant link between pyroptosis and glioma pathogenesis. Future studies encompassing basic experimental validation and clinical research are crucial to elucidate the precise mechanisms and impacts of pyroptosis in glioma.
2.6 Immunogenic cell deathPrior to the identification of ICD, apoptosis was typically regarded as a non-immunogenic process. The recent definitions by the Committee on Cell Death Nomenclature (NCCA) describe ICD as a regulated form of cell death that can activate host immune responses within an adaptive immune context. Unlike other forms of cell death, ICD is triggered by a narrow range of stimuli, including chemotherapy agents, viral infections, and various physical or chemical factors. These stimuli induce a cascade of damage-associated molecular patterns (DAMPs), subsequently eliciting an immune response (6).
Curcumin, a bioactive phenolic compound, exhibits antioxidant, anti-inflammatory, and antibacterial properties, and has been documented to modulate several cell death pathways. Research conducted by Zenghe Xiu et al. demonstrated that curcumin can potentiate ionizing radiation-induced death of glioma cells via activation of the endoplasmic reticulum (ER) stress PERK-eIF2α and IRE1α-XBP1 signaling pathways—a finding corroborated in murine models (86). Carbonic anhydrase IX (CAIX) is a tumor-associated cell surface glycoprotein that moderates tumor cell survival by influencing pH homeostasis. S4, a highly selective CAIX inhibitor, has shown substantial efficacy in breast and colorectal cancer models. Investigations by Jing Cui et al. revealed that S4 markedly diminishes glioma cell viability and induces both apoptosis and autophagy. Further analysis indicated that S4 promotes calreticulin exposure and the release of HMGB1 and HSP70/90, thereby activating the PERK-eIF2α and IRE1α-XBP1 pathways and facilitating DAMP release linked to ICD in glioma cells through the ER stress pathway (87). Recent studies assessed the potential of TNFAIP2 knockout in enhancing the therapeutic outcomes of PD-1 inhibitors. Both in vitro and in vivo experiments demonstrated that TNFAIP2 knockout increases surface expression of calreticulin (CALR), heat shock protein 70 (HSP70), and heat shock protein 90 (HSP90) in glioblastoma (GBM) cell lines, thereby inducing ICD. Consequently, TNFAIP2 knockout during PD-1 therapy could significantly bolster survival rates in glioma patients (88).
While molecular mechanisms underlying ICD have been extensively studied, the clinical implications of ICD in disease classification, treatment, and prognosis remain underexplored. Bioinformatics offers a robust platform to address these challenges. Jiayang Cai et al. identified 34 ICD-related genes, ultimately focusing on 12 key genes including IL17RA, IL1R1, EIF2AK3, CD4, PRF1, CXCR3, CD8A, BAX, PDIA3, CASP8, MYD88, and CASP1. They established associations with non-codeletion of 1p19q, higher WHO grades, wild-type IDH, and immunosuppressive tumor microenvironments (89). Additionally, Sun et al. analyzed 1,896 glioma samples across five databases, devising a risk score model based on 14 ICD-associated genes that independently predicts survival and response to immunotherapy in glioma patients (90).
2.7 Crosstalk between multiple RCDsDifferent modes of RCD, including apoptosis and necroptosis, have been extensively studied and elucidated. Apoptosis, characterized by its orderly and systematic nature, is vital for maintaining tissue homeostasis and eliminating damaged cells, thereby influencing numerous diseases and pathophysiological states. Under physiological conditions, apoptosis ensures the timely demise of cells through the meticulous regulation of both intrinsic and extrinsic signaling pathways (91). Glioma cells frequently demonstrate overexpression of anti-apoptotic proteins (e.g., Bcl-2, Bcl-xL) and deactivation of pro-apoptotic proteins (e.g., Bax, Caspase family). This apoptosis evasion mechanism not only enhances the survivability of tumor cells but also increases their resistance to conventional chemoradiotherapy and chemotherapy (92). Hence, restoring the normal apoptotic pathway is a pivotal research direction in anti-glioma therapies, with the reactivation of apoptosis presenting a promising strategy for effective tumor cell eradication. Necroptosis, a form of RCD that intersects characteristics of both apoptosis and necrosis, is primarily regulated by the receptor-interacting serine/threonine-protein kinases 1 and 3 (RIPK1 and RIPK3). In contrast to apoptosis, necroptosis resembles necrosis through the rupture of the cell membrane and the release of pro-inflammatory factors (93). Glioma cells often manipulate the necroptosis pathway to avert cell death, including the inhibition of RIPK1/RIPK3 activity and the prevention of downstream mixed lineage kinase domain-like protein (MLKL) translocation (94). The induction of necroptosis via exogenous signals such as tumor necrosis factor (TNF) or chemotherapeutic agents can lead to irreversible cell death in glioma cells, coupled with a potent inflammatory response that augments the immune system’s ability to target tumor cells. However, the excessive activation of necroptosis can also precipitate unnecessary tissue damage and inflammation, underscoring the necessity of precise regulation and the establishment of an optimal therapeutic window. Overall, a deeper understanding of necroptosis in glioma could pave the way for the development of innovative targeted therapies, thereby enhancing patient outcomes.
Tumors exhibit intricate crosstalk among various RCD pathways. Despite mechanistic and regulatory differences, these pathways can interact and influence one another under certain conditions, highlighting the importance of intercellular communication, especially as tumor cells develop mechanisms to evade drug-induced apoptosis in the context of targeted therapies.
Pyroptosis and apoptosis are primarily interconnected through the caspase family. The caspase-3/GSDME axis acts as a switch between these two forms of cell death, significantly impacting conditions like lung cancer and melanoma (95, 96). In infectious disease contexts, ZBP1 functions as a sensor for NLRP3 inflammasome activation triggered by influenza virus, paralleling the role of caspase-11 or caspase-4/5 in the non-classical LPS-induced pathway, intertwining with both pyroptosis and apoptosis. Additionally, ZBP1 is crucial in initiating PANoptosis in response to IAV (97).
The tumor suppressor gene TP53 mediates apoptosis and ferroptosis. TP53 upregulates numerous pro-apoptotic genes including BAX, PUMA, and NOXA, promoting mitochondrial-mediated apoptosis by inducing mitochondrial outer membrane permeabilization, releasing cytochrome c, and activating downstream caspases. TP53 also suppresses the SLC7A11 gene, which encodes a component of the system Xc^- transmembrane amino acid transporter responsible for importing glutathione precursor cysteine. Consequently, inhibition of SLC7A11 by TP53 reduces glutathione synthesis, leading to lipid peroxide accumulation and inducing ferroptosis (98). Ferroptosis is characterized by ROS generation and lipid peroxidation, processes that intersect with endoplasmic reticulum (ER) stress. Under ER stress, transcription factors such as IRE1, PERK, and ATF6 are activated, regulating apoptosis-related proteins like Bcl-2 and cytochrome c, thereby influencing the apoptosis pathway. Thus, modulating ferroptosis can also impact apoptosis (99).
Autophagy, particularly lipid autophagy and Beclin-1-mediated xCT degradation, can precipitate ferroptosis. Recent research has revealed that autophagy can induce ferroptosis through the regulation of intracellular ROS, LIP, and lipid peroxide levels, defined as autophagy-dependent ferroptosis (52). Studies by Eunhee Park et al. demonstrated that autophagy could be induced by ROS generated by erastin, an inducer of ferroptosis, with ferritin degradation and increased transferrin receptor 1 (TfR1) expression leading to ferroptosis. NRF2 plays a pivotal role in both ferroptosis and necroptosis; it can activate NLRP3 and AIM to induce necroptosis and exhibits specific functions in ferroptosis. Contrarily, NRF2 can also inhibit NLRP3 activation by reducing ROS levels, thereby blocking necroptosis (100, 101).
ICD is distinctive for its capacity to elicit an immune response. Although distinct from apoptosis, programmed necrosis, autophagy, and ferroptosis, these pathways can transition into ICD under specific conditions. Research on processes such as copper-induced cell death and aneuploid apoptosis in gliomas remains limited, with insufficient evidence to confirm their interaction with other RCD pathways. Collectively, the interrelationships among various RCD pathways are complex and tightly knit, forming an interactive network that regulates cell death and immune responses. Understanding these relationships is paramount for advancing treatments for tumors, autoimmune diseases, and infectious diseases.
3 The promise of RCD in targeted glioma therapy3.1 Potential application of autophagy-dependent cell death in glioma targeted therapyAutophagy-dependent cell death is a type of RCD that relies on autophagy mechanisms or components and plays a significant role in the central nervous system (28). Despite extensive research, there is still no definitive causal link between autophagy and cellular demise. The interplay between autophagy and various forms of cell death makes it challenging to distinguish them based solely on morphological observations throughout the cellular lifecycle. Therefore, here we mainly discuss the therapeutic concepts and methods based on inhibiting or accelerating the autophagy process to hinder tumor development.
The underlying mechanisms of autophagy that contribute to cellular death are notably distinct from those that promote cellular survival. These differences are primarily characterized by variations in the rate of autophagic flux, the duration and intensity of the involved pathways, as well as the nature and turnover rates of the substrates undergoing autophagic recycling and degradation. Such nuances play a crucial role in determining the balance between cell viability and death, thus representing potential focal points for innovative tumor therapies. A wealth of preclinical research has meticulously examined the function of autophagy in tumor treatment, employing two prevalent methodologies. The first method involves triggering autophagy through the administration of broad-spectrum anticancer agents, such as rapamycin, which antagonizes mTORC1 (102). The second strategy involves indirectly activating autophagy by selectively inhibiting specific drug targets, such as the inhibition of ERK in PDAC cell lines (103). Although the precise mechanism underlying modulation of autophagy remains elusive, the current understanding leans towards the notion that upregulation of autophagy confers a protective effect on tumor cells.
With the increasing understanding of autophagy, novel therapeutic agents targeting glioma cell autophagy have entered clinical trials. Chloroquine (CQ) and hydroxychloroquine (HCQ), known for their anti-malarial properties, have shown potential in inhibiting autophagy and enhancing the efficacy of DNA damage therapy. Previous studies investigated combination therapy with hydroxychloroquine for glioma by conducting phase I/II clinical trials to determine its efficacy and maximum tolerated dose (MTD). Despite effectively inhibiting autophagy, the clinical application of hydroxychloroquine is limited due to its toxicity at higher dosages (104). After that, while HCQ has been widely used in the treatment of malaria (105), primary progressive multiple sclerosis (106), IgA nephropathy (107), and even COVID-19 (108), it has rarely been utilized for glioma. However, in 2020, due to the COVID-19 pandemic and the urgent need for new glioma treatments, HCQ and CQ have regained attention, leading to the emergence of corresponding clinical trial studies. In a recent study by Inge Compter et al., the investigators examined the safety, pharmacokinetics, and MTD of a combination therapy consisting of chloroquine, RT, and daily TMZ administration in newly diagnosed glioblastoma patients. This novel therapeutic approach holds promise for the treatment of gliomas (109).
Furthermore, due to the gradual advancement of autophagy research and the rapid progress in bioinformatics in recent years, numerous studies have progressed to the stage of clinical trials. Mutations in the epidermal growth factor (EGF) have been implicated in gliomas’ resistance to conventional chemotherapy (110). Chloroquine, a potent autophagy inhibitor, impairs this survival mechanism by blocking the lysosomal degradation pathways activated by mutated epidermal growth factor receptor (EGFRvIII). This recently registered clinical study marks a new frontier in efforts to overcome therapeutic resistance within gliomas. A pioneering Phase I/II clinical trial, initiated in 2020, is investigating the combined efficacy of dabrafenib, trametinib, and hydroxychloroquine in the treatment of recurrent gliomas characterized by BRAF mutations. This trial specifically focuses on determining the extent of autophagy inhibition and its correlation with resistance profiles of gliomas, as well as the specific involvement of autophagic processes in the context of BRAF anomalies. Building on preclinical findings, delta-9-tetrahydrocannabinol and cannabidiol (CBD) have demonstrated their potential in tempering glioma proliferation and metastasis. These phytocannabinoids engage type 1 and type 2 cannabinoid receptors, triggering the endoplasmic reticulum stress response and subsequently inducing autophagy (111). Last year witnessed the initiation of a Phase I multicentric clinical trial in Spain dedicated to assessing the therapeutic safety of this novel combined modality. Phase I/II clinical trials are currently underway to investigate sphingoid modulators and the glioma autophagy inducer idroxioleic acid. These studies aim to determine the MTD of these drugs, evaluate their safety and initial efficacy, and shed light on their potential for anti-tumor treatment of glioma (112).
Unfortunately, numerous autophagy-related molecules possess a wide range of vital physiological roles beyond their established functions within the autophagic pathway. Autophagy-related genes (ATGs) exert their influence on various processes, both in healthy and pathological states, that go beyond their canonical duties as autophagy facilitators. For example, the ATG5-ATG12/ATG16L1 protein complex has demonstrated antiviral capabilities mediated by interferon-gamma—actions that differ from its involvement in autophagy (113, 114). Additionally, ATG7 is one of the core proteins of autophagy, but the ATG7 dependent non-autophagy pathway plays a key role in the regulation of neovascularization (115). While the classical autophagy pathway remains an essential aspect of ATG function, the non-autophagic capabilities demonstrated by these molecules cannot be disregarded. The multifaceted nature of autophagy-related molecules often complicates the understanding of their complex physiological interactions, presenting a significant challenge in unraveling their roles in clinical trials, which may contribute to suboptimal outcomes observed. Future research endeavors must adopt a broader perspective that is not confined to the solitary functions attributed to autophagy-related molecules. Enhancing our understanding of their versatile involvement in cell death regulation and the distinct disease-modulating processes mediated through autophagy-dependent death pathways in various neoplastic contexts will be crucial for advancing therapeutics.
3.2 Potential application of anoikis in glioma targeted therapyAnoikis, a distinct form of RCD triggered by cell detachment, holds significant importance in maintaining cellular homeostasis and microenvironmental stability. Mechanisms that activate resistance to anoikis hinder the induction of apoptosis in cells upon tumor initiation, impeding their ability to undergo cell death when detached. This phenomenon promotes the proliferation and migration of tumor cells. Consequently, inhibiting resistance to anoikis and restoring normal anoikis processes emerge as promising avenues for exploration in tumor research (Figure 3).
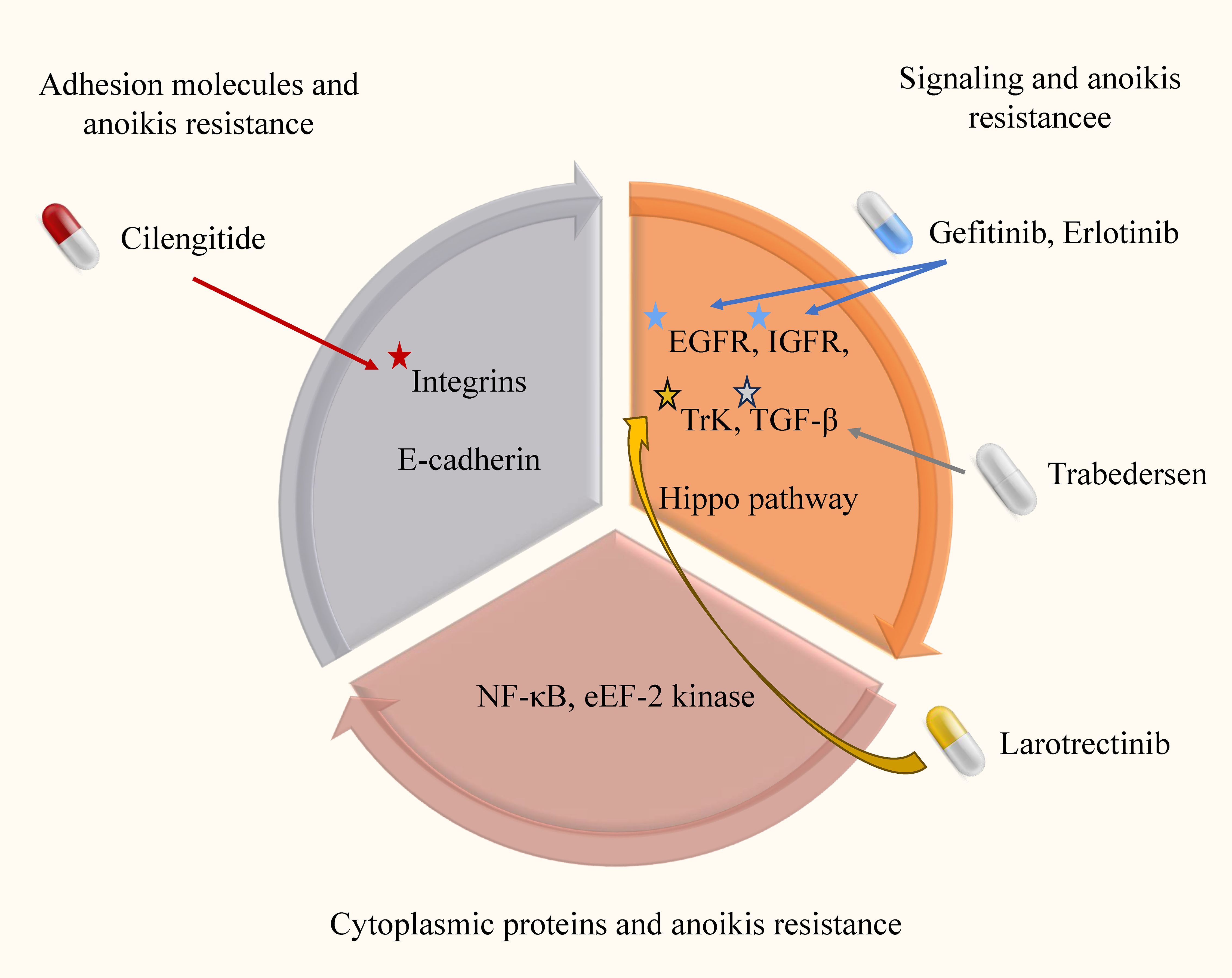
Figure 3. Anoikis resistance mechanism and targeted drugs in glioma. In this image, we elucidate the intricate anoikis-resistance pathways operative in glioma, along with their consequential clinical implications. Anoikis, which is inherently designed to eliminate impaired cells, is crucial for maintaining cellular equilibrium within the microenvironment. However, in gliomas, a disrupted resistance to anoikis emerges, enabling cancerous cells to evade this regulatory process and disrupting the natural course of malignant cell clearance.
Despite the inherent radiation resistance of gliomas, RT remains a standard treatment in the context of tumor drug resistance (116). In a study of prostate cancer, Erk and PIK-AKT signaling enabled isolated tumor cells to acquire resistance related to anoikis, leading to drug resistance (117). In glioma, the commonly used alkylating agent TMZ is an essential component of adjuvant chemotherapy. However, recent research has demonstrated that astrocytes resistant to anoikis exhibit robust resistance to TMZ, resulting in minimal reduction in cell viability and unaffected colony formation (118). These findings suggest a potential association between induction of anoikis and drug resistance in glioma.
Considering the potential correlation between anoikis and the development of drug resistance in RT and chemotherapy, targeted therapy against anoikis demonstrates promise as an ideal adjunctive strategy in glioma treatment. Exploring the molecular regulation of anoikis may provide novel insights into prognostic assessment in glioma. For instance, a recent study developed a prognostic model for low-grade glioma (LGG) based on the ANRG and LGG subtypes (119). Another investigation employed differentially expressed anoikis-related genes (ARG) derived from GeneCards to construct a prognostic ARG model for glioma, revealing that patients in the high-risk group exhibit a worse prognosis (61). These findings collectively underscore the clinical significance of anoikis in glioma treatment and prognosis, despite the need for further mechanistic investigations. Continued research in this area will undoubtedly contribute to advancements in glioma therapeutics.
3.3 Potential application of ferroptosis in glioma targeted therapyWith the increasing understanding of ferroptosis, researchers have initiated investigations into its potential in tumor therapy. Multiple studies have demonstrated that specific anti-tumor drugs can induce ferroptosis, highlighting its potential in glioma treatment.
Recent findings suggest a close association between ferroptosis and treatment resistance in targeting tumor cells. Viswanathan et al. demonstrated that the lipid peroxidase pathway, mediated by GPX4 which can partially suppress ferroptosis, regulates the development of highly resistant mesenchymal cells (120). Another study revealed that depleting immunosuppressive cells in the tumor microenvironment can reverse drug resistance through ferroptosis (121). These findings underscore the potential of ferroptosis as an effective adjunct therapy against glioma resistance.
Extensive research has been sparked into the potential induction of ferroptosis in tumor treatment, with several clinically approved drugs (e.g., sorafenib and artesunate) demonstrated to effectively induce ferroptosis (122). The integration of nanomaterials with targeted ferroptosis has garnered significant attention in the field of tumor treatment research. Nano-based drug delivery systems offer advantages such as enhanced stability, improved availability, minimal side effects, and robust targeting capabilities. Chen et al. developed trehalose-loaded nanoparticles that combined GSH consumption-induced ferroptosis with trehalose-induced autophagy, demonstrating the potential of nanaterial design in developing ferroptosis-induced nanomedicine (123). While most nanoscale research on ferroptosis has focused on breast and liver cancer, this approach is expected to become a future treatment strategy for glioma. It also demonstrates the importance of interconnection between different RCD pathways.
The increasing recognition of the significance of ferroptosis in the tumor microenvironment and its potential as a prognostic predictor has led to studies investigating its clinical relevance. Zhuo et al. conducted a study examining genetic markers associated with ferroptosis and found that these markers could potentially serve as prognostic indicators for glioma patients (124). Similarly, Wan RJ et al. identified differential expression of ferroptosis-related genes between glioma patients and non-patients, suggesting the possibility of using these genes as a new standard for prognostic evaluation in the future (125). These findings highlight the significance of comprehending the role of ferroptosis in the tumor microenvironment and its potential as a prognostic marker for glioma patients. Further research is necessary to validate and expand upon these findings, ultimately advancing prognostic prediction in glioma.
3.4 Potential application of cuproptosis in glioma targeted therapyPerturbations in serum and tumor tissue copper levels have been observed in patients with malignant tumors such as lung cancer (126) and breast cancer (127), suggesting alterations in copper homeostasis that may contribute to tumor progression, invasiveness, or drug resistance. Although experimental research on cuproptosis is still in its early stages, it is undeniable that cuproptosis holds promising potential as a therapeutic target for tumor treatment.
Pertinent findings from several studies have established a correlation between cuproptosis markers and the prognosis of glioma. In research conducted by Li Wang and colleagues, notably elevated expression levels of cuproptosis-related genes were observed in the high-risk group of glioma patients as compared to the low-risk group. Moreover, the researchers developed a risk model grounded on long non-coding RNA (lncRNA) associated with cuproptosis, which partially reflects its prognostic significance in glioma (79). However, additional experimental validation is necessary to corroborate the prognostic implications of cuproptosis-related genes in this disorder.
In our review of tumors, we have discovered that certain metal ions, namely cadmium (128), chromium (129), and bismuth (130), possess carcinogenic properties to some extent (Table 2). Recent research suggests that their mechanism of action may be closely linked to ferroptosis, cuproptosis, and autophagy. Ashish Tyagi et al. conducted mouse experiments and found that chronic exposure to cadmium consistently activates the NOX1 complex, leading to the generation of ROS and endoplasmic reticulum (ER) stress in cells. This activation subsequently results in defective autophagy (131). In a separate study conducted by Caijun Zhao et al., it was discovered that cadmium-induced ER stress mediates the activation of autophagy, which leads to ferroptosis in renal tubular epithelial cells (128). The potential therapeutic application of these metal ions in disease treatment is a topic of discussion, for instance, bismuth has been widely used for the eradication of H. pylori infection (132). It is important to note that the therapeutic and side effects of metal ions are often dose-dependent and influenced by the method of administration. Currently, metal ions have not been utilized in tumor therapy, and further research is necessary to explore their potential in this field.
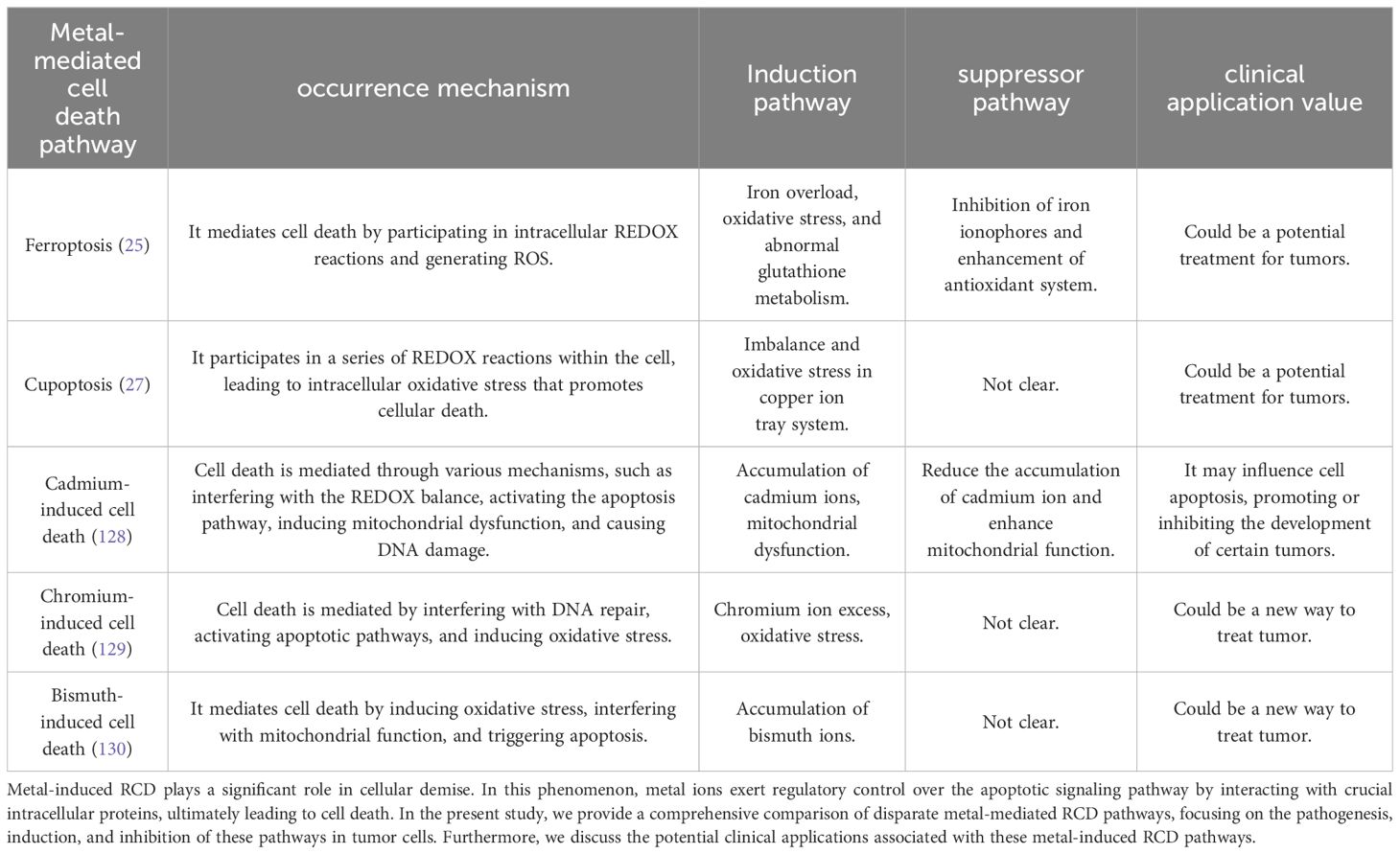
Table 2. Comparative analysis of metal-mediated RCD pathways.
3.5 Potential application of pyroptosis in glioma targeted therapyThe relationship between pyroptosis and tumor dynamics, particularly in glioma, is intricate and multifaceted. On one hand, as a significant form of RCD, pyroptosis can effectively inhibit tumor initiation and progression. Conversely, the inflammation and immune responses triggered by pyroptosis release various inflammatory mediators and cytokines, such as interleukin-1β and interleukin-8, which might promote tumor growth, modify the tumor microenvironment, and contribute to drug resistance (133).
Current research endeavors aim to harness pyroptosis to enhance glioma treatment. For instance, Li-Wen Ren utilized weighted gene co-expression network analysis (WGCNA) on data from The Cancer Genome Atlas (TCGA) to identify key genes in glioblastoma (GBM). Using the connectivity map (CMAP) platform, three substances—flubendazole, mebendazole, and fenbendazole—were identified as potential anti-glioma agents. These compounds were shown to induce pyroptosis in GBM cells via the NF-κB/NLRP3/GSDMD pathway and also trigger mitochondria-dependent apoptosis in nude mice models (134). In another study, Wenpeng Zhao and colleagues conducted high-throughput screening of 2,718 compounds from approved drug and clinical compound libraries. They discovered that the second-generation small molecule polyCDK inhibitor AT7519 holds promise as a GBM therapy. AT7519 notably inhibited glioma cell viability and proliferation in a dose-dependent manner and induced pyroptosis through caspase-3-mediated cleavage of gasdermin E (GSDME) (135). Meanwhile, Chinese researchers have explored traditional Chinese medicine formulations, identifying Xihuang Pill as a noteworthy candidate. This traditional preparation promotes the pyroptosis of glioma cells via the POU4F1/STAT3 axis and effectively halts glioma proliferation, although its precise mechanisms and clinical applicability warrant further investigation (136). Despite these promising approaches, translating them into effective clinical treatments faces substantial challenges. The glioma microenvironment is exceptionally complex and exhibits significant heterogeneity in treatment response. Furthermore, researchers must address issues of selectivity, drug delivery, side effects, and resistance associated with pyroptosis-inducing agents.
In summary, while pyroptosis offers potential therapeutic avenues for glioma, extensive research is necessary to overcome the
留言 (0)